 
Alternatively, Roman numerals can be written as a right-hand superscript, 7 e.g., Cr VI, or as Arabic numerals preceded by the appropriate charge sign, 2- 3 e.g., Cr(+6) and Cr +6, or by placing the oxidation number exactly above the appropriate chemical symbols (see Table 2). An oxidation number of zero may be represented by the numeral 0, but this is not usually shown. An oxidation number so written is always non-negative unless the minus sign is explicitly used (the positive sign is never used). Note that there is no space between chemical symbol and parenthesis. 4 The oxidation numbers are denoted by the Roman numerals in parantheses after the element name, e.g., Cr(VI). 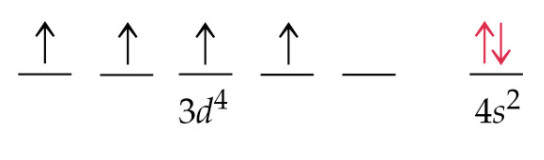
1, 3 In other words, it is the hypothetical charge an atom would have if all bonds to atoms of different elements were treated as ionic. (4) The oxidation state is defined as the charge of an atom in a molecule after ionic approximation of its heteronuclear bonds. On the other hand, the concept of oxidation state (oxidation number) is a formalism. It relates to all areas of science in which the chemistry of aqueous solutions comes to the fore, although the motivation for writing this note comes from the field of corrosion and corrosion protection of metals. Further, its purpose is to bridge the gap between the academic and industrial backgrounds in presenting the established results of research to a wide audience. The present discussion is aimed to attract the attention of researchers from various fields of science to prevent further use of misleading notations and interpretation. It is thus of great interest to produce scientifically accurate papers that obey the IUPAC (1) notation. The auto-detrimental effect caused by the accumulation of incorrect records is evident in the literature. Another problem may be simply an insufficient level of accuracy, which is not acceptable in scientific records. This is obviously related, not only to the authors of the scientific papers, but also to the reviewers and editors, who are responsible for the correct use of scientific terms. One of the main contributing factors to these mistakes or even misunderstandings may be related to the fact that the corrosion researchers have different backgrounds, not necessarily the most frequent being in chemistry, but in other fields important in corrosion research, such as material science, metallurgy, engineering, and physics. Once published, these incorrect entries persist, being cited and spread further without being recognized as incorrect. This can be seen not only in high-ranking scientific journals concerned with the field, but also in the records of international conferences presented to eminent audiences. Specifically, incorrect notations and even scientific misinterpretations associated with the chemical terms “charge” and “oxidation state” are noted. The lowering of the oxidation state of a metal by the flow of negative charge from its coordinated ligands is reflected in the reduction of the C-O stretching frequencies.More and more incorrect use of chemical nomenclature has appeared in the last decades in the scientific literature concerning the field of corrosion. Infrared spectra are useful because carbonyl frequencies are easily detectable. Since accumulation of superfluous electrons on a low oxidation state metal atom is prevented, back-donation leads to the stabilization of the M-C bond.įig 6.15 - Back donation in metal carbonyls.Ī rise in the order of the metal - carbon bond is reflected in the increase of the M-C, and decrease of the C-O, stretching frequencies in vibrational spectra. \) orbital from the filled metal d orbital is called back donation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
